The Patient Navigation Program of the Musella Foundation and the Promising Pathway Act
While completing the procedure for the transformation of Glioblastoma.it into the voluntary association “Glioblastoma.it ODV”, I have found a very interesting video the Musella Foundation that explains some details of the operations of the Patient Navigation Program and describes the Promising Pathway Act that is being proposed for approval by the US health system.
In practice it is known that (some retrospective articles have also been published and we have already talked about them on this site) those who participate in some clinical trials do better on average. This is due not necessarily to the Clinical trial result but maybe to the fact that these patients are receiving more attention or simply because their emotional stated is better just because by participating in a clinical trial they have hope. In practice we don’t know why but this is how it works.
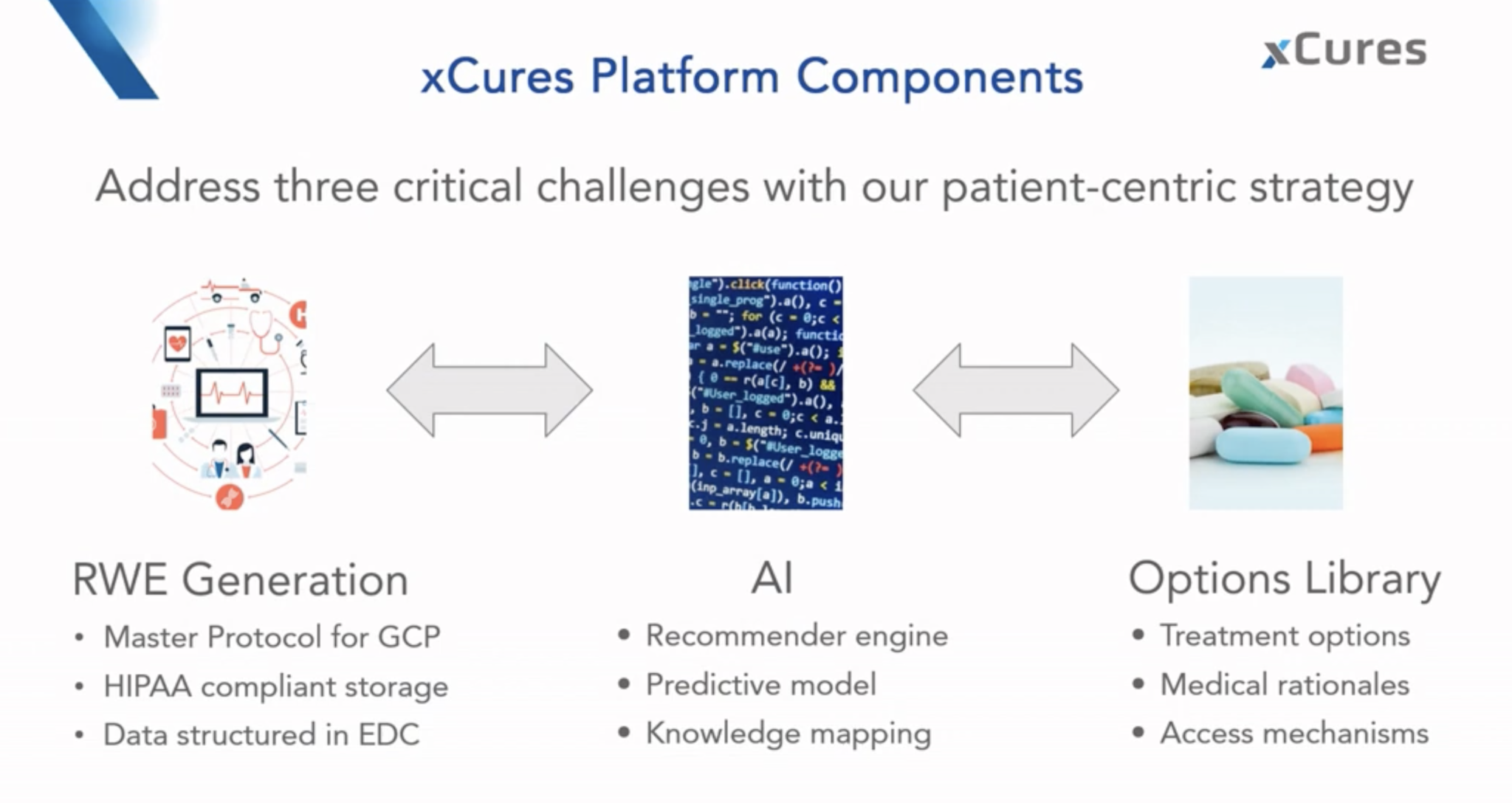
Now, participating in clinical trials is not easy … there are several constraints or the timing is not right etc etc. How can we take care of patients who are not participating in clinical trials? This is why the Patient Navigation Program was launched by the Musella Foundation in collaboration with the xCures company. The following figure describes xCure platform which is the core of the patient navigation program. In practice, patients sign in and provide information about their glioblastoma and the treatments received. A team of specialists suggests treatment options from an always updated treatment option library with the support of an artificial intelligence system.
I am always convinced of the importance of carrying out the Glioblastoma Navigator project that I dave already described. Basically this project aims to do many things that are part of the Patient Navigation Program. There is certainly a great need of support from medical specialists that I invite until now to get in touch with us if they are interested to collaborate.
Coming to the Promising Pathway Act. In summary, this law proposal establishes that for serious pathologies like glioblastoma multiforme the patients can try experimental treatments that have proven to be relatively safe (for example by passing the first phases of a clinical trial) and to provide positive therapeutic effects. These patients have to be followed and have to be enrolled in a database in order to be able to trace the effects of the their treatments and provide useful information to the healthcare system. Insurance cannot refuse to pay the treatment due to the fact that the treatments are considered experimental and the patients and their relatives cannot take legal action against doctors and pharmaceutical companies if the treatments fail to produce the desired effects. It seams to me this is an important law with an important potential impact on glioblastoma multiforme patients.
That’s all for today and remember the CUSP-ND fundraising campaign: trial to beat Glioblastoma in memory of Emanuele!